CLINICAL TRIAL LOGISTICS in JAPAN
GREEN8 is a specialized bio-medical logistics company in Japan that handles pharmaceutical, biological specimen and cell transportation. In clinical trial drug transportation, GREEN8 Japan provide all the necessary services for quality management of investigational drugs, including temperature control, preparation of transport materials, packaging, document processing, and verification tasks, all in a one-stop solution. GREEN8 also handles clinical logistics with overseas network.
In clinical trial drug transportation, GREEN8 Japan utilize dry shipper tanks capable of transporting at temperatures below -150°C, ensuring transportation while maintaining ultra-low temperature conditions. Additionally, we have a 24/7 call center to address emergency trial needs in Japan.
In addition to providing full logistics services, GREEN8 Japan has bilingual trained staff to conduct clinical trial coordination smoothly in Japan and to coordinate with English speaking sponsors, assisting the non-Japanese clients to navigate the complicated Japan requirements and Japan standards and expectations.
When choosing a clinical trial logistics company in Japan, it’s important to consider the following points:
Quality management system
International transportation capabilities
Emergency transportation response
Staff with troubleshooting skills available 24/7 in Japan.
GREEN8 covers all these aspects!
In clinical trial transportation, we transport investigational drugs and specimens collected from patients (such as blood, urine, tissue, cells, DNA, placenta, etc.).
GREEN8 : Clinical Drug Supply Chain Services in Japan
GREEN8 offers temperature-controlled pharmaceutical transportation services throughout Japan. We own our in-house GMP depo capable of temperature management, as well as required license to handle clinical drugs in Japan. We provide the following services:
Clinical Trial Drugs and Specimens Distribution in Japan
Transportation of investigational drugs, placebos, pre-trial drugs, patient specimens, and collection of used investigational drugs, signing for receipts, integration with clinical trial management systems.
Temperature-Controlled Logistics in Japan
Storage, Packaging, and Shipping in Japan
Compliance with GxP
International Trial Support
Handling Administrative Tasks
Flexible Logistics Network Utilizing Our Own Depots
From Clinical Trial Drug Storage Facilities (Depots) to Trial Implementation Sites (Medical Institutions)
Transportation from Investigational Medical Products (IMP/IP) storage facilities (depots) to medical institutions is a crucial phase in clinical trial drug distribution. With stringent quality control measures in place, delivery services are particularly vital within the realm of Investigational Medical Products (IMP/IP) logistics. At GREEN8, we ensure the stable preservation of investigational drug integrity while reliably and swiftly delivering them to medical institutions, ensuring appropriate provision to patients.


From Clinical Trial Drug Storage Facilities (Depots) to Patients' Homes
Transportation of Investigational Medical Products (IMP/IP) to patients’ homes is also a significant component. With nationwide distribution capabilities, Green8 provides assurance, allowing you to entrust us with confidence.
GREEN8: Temperature-Controlled Logistics in Japan
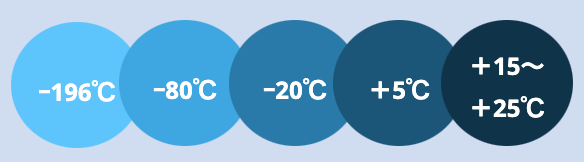
Transportation and storage across 5 temperature zones are feasible.
Depending on the characteristics of clinical trial drugs and specimens, transportation and storage are achievable across five distinct temperature zones. This flexibility ensures the maintenance of investigational drug stability and reliability in providing to patients.
Preparation of temperature-controlled Containers
Proper temperature management is essential for the transportation of clinical trial drugs and patient specimens. The preparation of refrigerated containers is integral to this process, as it is a necessary measure to maintain the quality of investigational drugs and specimens. We can provide validated temperature-controlled containers to ensure the preservation of integrity. Additionally, temperature loggers can also be provided simultaneously.

Items for Distribution
Clinical Trial Specimens
At Green8, we have GMP warehouses and provide transportation compliant with GDP. We have a track record of transporting various specimens such as blood, plasma, urine, tissues, cells, DNA, and more. Transportation of infectious substances is also feasible. Under strict quality control, clinical trial specimens are collected safely and efficiently transported to their destination. The collection and transportation of clinical trial specimens are essential processes to ensure the reliability and validity of trial results.
Investigational Medical Products (IMP/IP)
The refrigerated transportation and collection of Investigational Medical Products (IMP/IP), including test drugs and comparator drugs, require advanced refrigeration technology and specialized procedures. Investigational Medical Products (IMP/IP) needed for patient treatment are delivered safely and reliably, and used such clinical trial drugs are appropriately collected and disposed of. This process is integral to quality management.

Placebo
The refrigerated transportation and collection of placebos are conducted with the same level of caution as clinical trial drugs. Placebos are transported under precise temperature control to ensure the reliability of the trial. The collection of placebos is crucial for maintaining data accuracy and is carried out based on strict protocols.
Pre-Trial Drugs
The transportation and collection of pre-trial drugs are carefully planned from the consideration stage. To ensure smooth progress of the trial, the process of providing and collecting pre-trial drugs is efficient and reliable.

Used Clinical Trial Drugs (Collection and Disposal)
When clinical trial drugs are no longer needed, they are safely collected and disposed of according to appropriate procedures to minimize environmental impact. The collection of used clinical trial drugs not only contributes to environmental preservation but also forms part of proper pharmaceutical management.
Signing for Receipt of Clinical Trial Drugs (Preparation of POD)
In the transportation of clinical trial drugs, it is essential to prepare a sign-off sheet for the receipt of clinical trial drugs (POD). This procedure ensures that the accurate receipt of clinical trial drugs is documented, ensuring quality management and traceability.

Integration with IRT (Interactive Response Technology)
Integration with IRT (Interactive Response Technology) is indispensable for the transportation and collection of clinical trial drugs. This enables real-time data management, enhancing the efficiency and reliability of the entire trial process.
Green8's Clinical Trial Drug Storage Facility (Depot)
Equipped with state-of-the-art facilities capable of storage across 5 temperature zones, Green8’s depot enables temporary and medium-to-long-term storage of clinical trial drugs and specimens. With thorough sanitation measures and comprehensive security protocols, coupled with a zoning system across two buildings, we ensure a safe storage environment by segregating storage classifications for each specimen.


Packaging, Storage, and Shipping of Clinical Trial Drugs
Green8 also undertakes packaging, storage, and shipping operations based on GMP procedures for pharmaceutical manufacturing (packaging, labeling, storage classifications). While maintaining the quality of clinical trial drugs, we provide integrated services for transportation and storage.
Transportation Arrangements and Inventory Management of Clinical Trial Drugs
In international clinical trial projects, we can aggregate specimens collected at Green8’s storage facilities, and arrange for regular shipments of specimens domestically and internationally. We also offer assistance in coordinating delivery arrangements from end-users to their specified destinations. Additionally, we handle various export and import procedures for overseas research institutions, procurement sources, and distributors. We also provide comprehensive inventory management services, including the complex inventory management associated with the inbound and outbound movements from the depot.

Compliance with GxP
Expertise in GMP, GDP, and GCP Regulations
Specialized staff knowledgeable in various regulations, including the Pharmaceutical Affairs Law (GMP Regulations concerning pharmaceutical quality management), Good Distribution Practice (GDP Guidelines for appropriate distribution of pharmaceuticals), and GCP Regulations.
In clinical trial transportation, quality management is paramount. Therefore, rules for clinical trials are established in regulations such as the GCP Regulations. Additionally, if clinical trial drugs fall under hazardous substances, appropriate measures must be taken based on their classification.
At Green8, we have implemented a quality management system in compliance with GCP/GDP regulations. Furthermore, we are equipped to handle pharmaceuticals classified as hazardous substances under the Fire Service Act, including their handling, storage, and transportation. We also accommodate pharmaceuticals classified as poisonous or deleterious substances (transportation, storage, handling).


Training for Specialized Staff
At Green8, we conduct monthly basic education sessions for all staff involved in bio-medical transportation. These sessions cover specialized knowledge required in the medical industry, internal SOPs (Standard Operating Procedures) at Green8, as well as compliance regulations. Following the training, comprehension tests are conducted to ensure continuous improvement and advancement of our specialized staff.
After education on our mission, medical industry rules, internal SOPs, and compliance, comprehension tests are conducted to assess understanding.
Additionally, we provide procedural training and task instructions tailored to each staff member as needed for specific projects.
Prevention Measures for Human Error
At Green8, we assign two dedicated personnel to each project. This two-person system enables us to utilize the function of double-checking, reducing the occurrence of human errors. It also prevents the reliance on individual expertise, allowing for increased efficiency and quality improvement in our operations. This setup also ensures a backup system is in place for seamless operations.


Development of Procedures and Legal Compliance Seminars
At Green8, we have established internal SOPs (Standard Operating Procedures) in accordance with GCP/GDP/GMP standards. We continuously update our SOPs to ensure they remain current with industry best practices. Additionally, we offer the creation of project-specific SOPs as needed.
Furthermore, we conduct seminars on topics such as “Key Points and Practical Considerations for GDP Compliance in Pharmaceutical Transportation and Storage” and “Considerations for Clinical Trial Drug Transportation.” These seminars serve as part of our internal education and training program, and we welcome anyone interested in participating to contact us for more information.
International Clinical Trial Support
Export and Import of Clinical Trial Drugs (International Transportation)
The export and import of clinical trial drugs require international cooperation and collaboration. Through collaboration with overseas supply chains, clinical trial drugs can be safely and swiftly transported internationally, facilitating the smooth progress of international clinical trial processes.


Capability for International Clinical Trials and Bilingual Trial Coordinators
In the clinical trial process, bilingual trial coordinators capable of handling international trials are essential. By accommodating different cultures and languages, international trials can proceed smoothly, and clear communication can be achieved for patients and stakeholders.

