Cryo strage solution
- What is Egg Freezing or oocyte cryopreservation?
- GREEN8's Cryopreserved Storage Services
GREEN8 Japan supports your life plans by storing your reproductive cells in optimal environment. GREEN8 has been transporting human cells, embryos, oocytes and sperms between medical clinics for many years.

GREEN8 Japan transports more than 100 cryo shipments per year. Our transportation services have earned the trust of clinics, hospitals and laboratories in Japan and other countries.
As a professional in temperature-controlled transportation domestically in Japan as well as international shipments, GREEN8 handles and stores cryo cells with the utmost care.
GREEN8’s GDP/GMP storage depo is located in Yonezawa city, Yamagata prefecture in Japan. The risk of an earthquake of intensity 6 or higher within the next 30 years is less than 0.3-3% in Yonezawa, compared to the high risk of 30-100% of Japan metropolitan area. The geological layer is different from the Pacific Ocean plate of Tohoku region. The geographically stable ground of Yonezawa is proven to be low for risk of disasters due to mega earthquakes as there were no casualties in the Great East Japan Earthquake of 2011.
In addition, GREEN8’s storage facility is outstanding and meets the global standard of GDP/GMP guidelines for handling biological substances and pharmaceutical products. The global standards related to pharmaceuticals, which require delicate handling, demand high quality in various specifications, such as temperature control and security measures are met at GREEN8 storage warehouses. A temperature monitoring system is also in place, and alerts are issued in the event of temperature deviations.The number of people who can enter and leave the room is also restricted. Surveillance CCTV cameras are also in operation 24 hours of monitoring.
Please feel free to advantage of GREEN8’s world class reefer storage services in Japan.
- Oocyte cryopreservation program in Japan


- GREEN8's risk reduction measures
GREEN8 facility is constructed with earthquake resistant structure as well as measures to withstand other natural disasters such as typhoons and flooding. While the geographic location of the GREEN8 Japan’s storage facility is in a low-risk terrain (according to National Research Institute for Earth Science and Disaster Prevention), GREEN8 facility is also equipped with electric power generator that could support the temperature-controlled facility for 7 days in case of a power outage.
Reference: J-SHIS MAP (National Research Institute for Earth Science and Disaster Prevention) https://www.j-shis.bosai.go.jp/map/
To overcome mix ups as well as possible human error in biological specimen or any stored samples, GREEN8 keeps tracks of each sample through digital barcode management system. Also to avoid human error, SOPs are in place for trained personnel to double check each entry and exit.
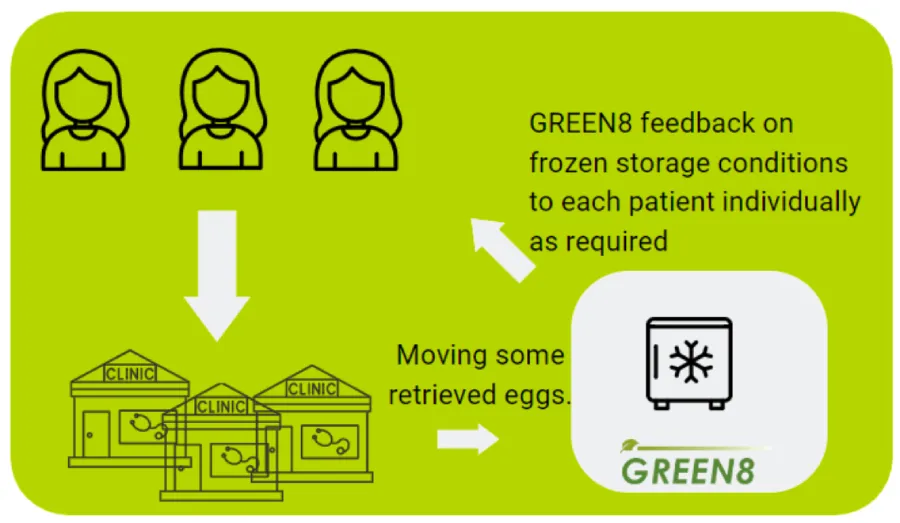
- Flow of use (egg freezing)
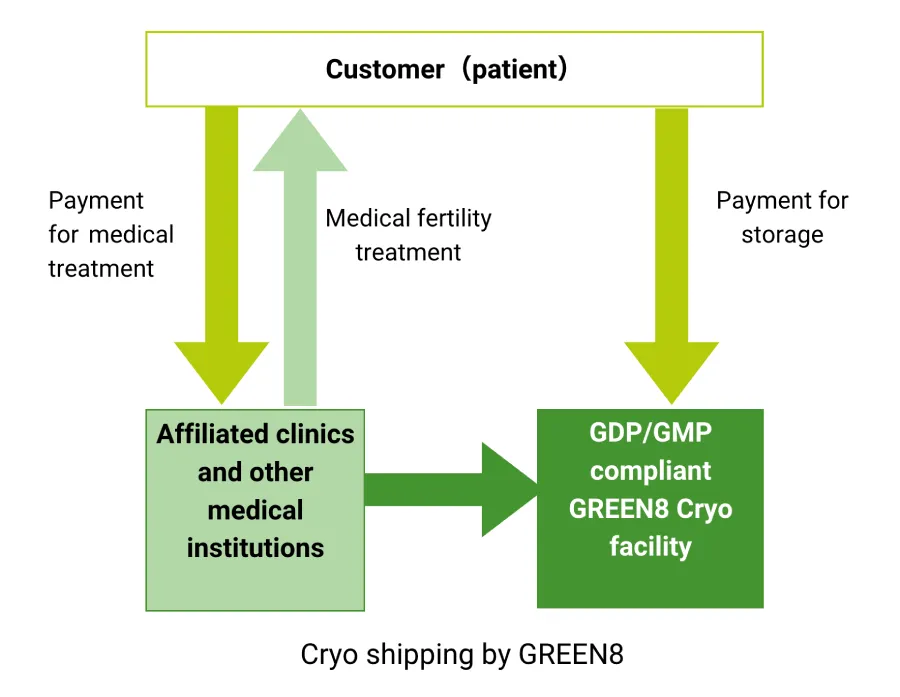
- About GREEN8's storage facilities
GREEN8 cryo facility is located at the its GDP/GMP compliant facility in Yonezawa, Japan. The facility is registered at “Yonezawa Repro-Medicine Center” in which capable of handling reproductive, pharmaceutical , biological and scientific substances and material.
Please contact us for not only frozen oocytes, embryos and sperm short term and long term storage, but also any bio-pharma related storage and distribution needs in Japan.
GREEN8 Japan facility provides 5 different controlled temperature zones, in which each zone is equipped with 24x7 temperature monitoring system. The system is set to sound an alert if the temperature deviates, and logs and records are available upon client request.
GREEN8 Japan facility complies with international and Japan national standards of GMP (Good Manufacturing Practice) and GDP (Good Distribution Practice) standards for storing and handling pharmaceutical products.
- Q&A

