GMP Pharma Depo Capabilities
Pharmaceutical Raw Material Supply Chain Depot ( GMP and GDP compliant storage facility)
Since 2013 GREEN8 has been engaged in international temperature-controlled bio-pharmaceutical logistics services, handling life science-related products such as pharmaceuticals, biologicals, Dangerous Goods, and medical equipment. In addition to transportation, we operate a GMP and GDP compliant storage facility in Japan in which utilizes our accumulated expertise in life science field, and can provide total logistics support from the R&D site to the marketplace for pharmaceutical raw material, API, intermediates, and clinical trial supply chain.
GREEN8 Japan’s Pharma Supply Chain Support Services
GREEN8 coordinates optimal and comprehensive logistics services for clients as an asset-based "3PL (3rd Party Logistics)" partner in Japan. 3PL is an agent that undertakes the entire logistics recruitment for the client, and performs all logistics-related tasks such as domestic transportation, storage, inventory management, documentation, receiving, shipping, re-packing, re-labeling and import/export in the most efficient manner. GREEN8 as your 3PL agent in Japan undertakes all logistics-related operations such as Japan domestic transportation, storage, inventory management, documentation, shipping/receiving, import/export, etc. on behalf of the shipper in the most efficient manner. Through our own inhouse GDP-compliant warehouse, GREEN8 supports the supply chain related to drug discovery through commercial distribution in Japan.
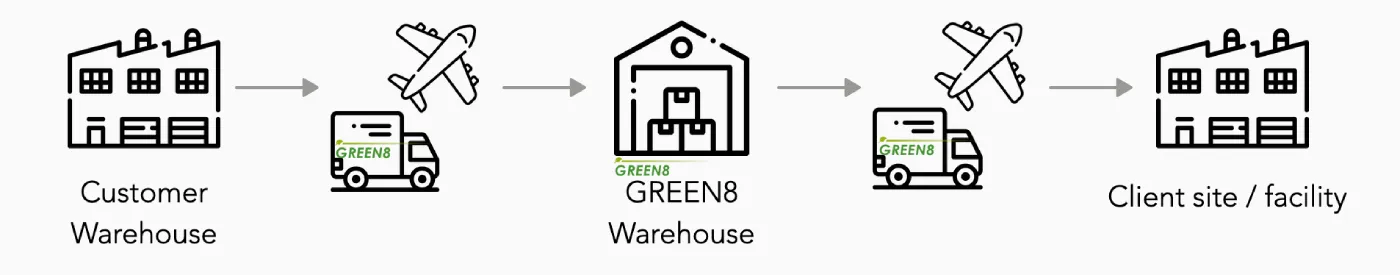
■Capabilities for storing Pharmaceutical, API and Bulk Drug Substances
GREEN8 Japan is capable of storage and distribution of Active Pharmaceutical Ingredients (API) and Bulk Drug Substances in Japan. This includes short-term as well as long term storage and distribution of pharmaceutical intermediates and API which require GMP and GDP requirements, as well as temperatures-controlled warehousing and distribution in Japan. Flexible temperature-controlled zones are provided in 4 main ranges for pharmaceutical and bulk drug substances available for CRT 15-25℃, chilled 2-8℃, Frozen minus 15-20℃, as well as deep freezers at minus 75-80℃ ranges. Secure your business continuity in Japan with sourcing required logistics solutions by GREEN8 Japan.
■Complaisance and Safety measures
Safety and compliance are essential core of GREEN8 services and solutions. Complying with relevant Japan laws in the storage, handling, transport, and distribution of pharmaceutical material and dangerous goods is getting more and more important. Our GMP and GDP certified facility is certified by Japanese regulatory agents, and compliance measures are implemented through internal SOPs as well as regular training sessions for staffs and personnel.
■Storing frozen pathological and scientific specimens
GREEN8 Japan’s GMP and GDP depo facility is equipped with storing frozen pathological and scientific specimens which are required to be kept at deep freezers at -80 or -170 degree Celsius for long-term. By subcontracting proper storage of valuable frozen research specimens to GDP compliant storage facility, R&D and academic laboratories can reduce their costs related to in-house storage, and more flexibility is available for actual medical and scientific research. Depending on client needs, each frozen specimens can be stored in separate racks, zones, or buildings depending on each substance’s hazardous and safety levels.
■Clinical Trial Drug Depot and Clinical Supply Chain Services
GREEN8 Japan facility is capable of storage of clinical drugs for international trial as well as Japan domestic clinical projects. This includes storage and distribution of clinical drug products from arrival airport, to GREEN8 Japan’s drug depo, and then distribution to each investigator sites in Japan through GDP standards. Pickup and storage of patient’s biological specimen is also possible through validated packing solutions for different temperature ranges. Contact us for customized clinical trial logistics that fits your SOPs in Japan.
■Business Continuity Plan (BCP) measures
In Japan, where earthquakes and other major natural disasters can strike at anytime and anywhere, most pharma storage facilities are concentrated in the Tokyo and Osaka metropolitan areas. In recent years, the importance of decentralized storage of R&D and commercial pharmaceutical products are considered an essential measure for business continuity plan (BCP) for sustainability business activity in Japan. GREEN8 facility is located in a low-risk North East area of Japan which is also accessible easily from Tokyo metropolitan area for fast and smooth supply chain service.
GREEN8 Japan Storage Depo
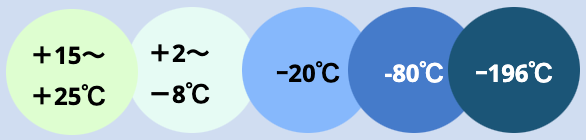
GREEN8 Japan storage depo is equipped with temperature-controlled facility in which storage of biological specimens, as well as pharmaceutical raw materials, intermediates, and APIs are possible in 5 different controlled temperature chambers through most advanced 24 hours monitoring system.
GREEN8 provides high quality services in compliance with GMP (Good Manufacturing Practice/ Good Manufacturing Practice and Quality Control Standards for Pharmaceuticals) ordinances, as well as GDP (Good Distribution Practice/ Good Distribution Practice for Pharmaceuticals) standards. We provide temperature monitoring and traceability assurance through our trained and professional staff.
Japan local and federal regulation on handling, storing and distribution of chemical and explosive substances is strictly regulated by Japanese law. In addition to capabilities in handling IATA DGR regulated substances, GREEN8 Japan is competent in storage and distribution of DG products controlled as Class IV (flammable liquids) under Japanese Fires Services Act. Prevent explosions or fires by storing your chemical material below its flash point, and delegate GREEN8 Japan to properly store your chemical substances with low boiling points. GREEN8 place emphasis on compliance and safety in accordance to Japanese as well as international standards.
Temperature control facility
■Controlled temperature environment
GREEN8 Japan’s facility provides five(5) different temperature controlled zones: CRT (+15~+25C), refrigerated (+2~+8C), frozen (-20C), deep freezer(-80C), and cryo tanks(-170-190C). In addition to medical deep freezers, Cryo storage is possible through liquid nitrogen storage tanks with monitoring and backed emergency systems.
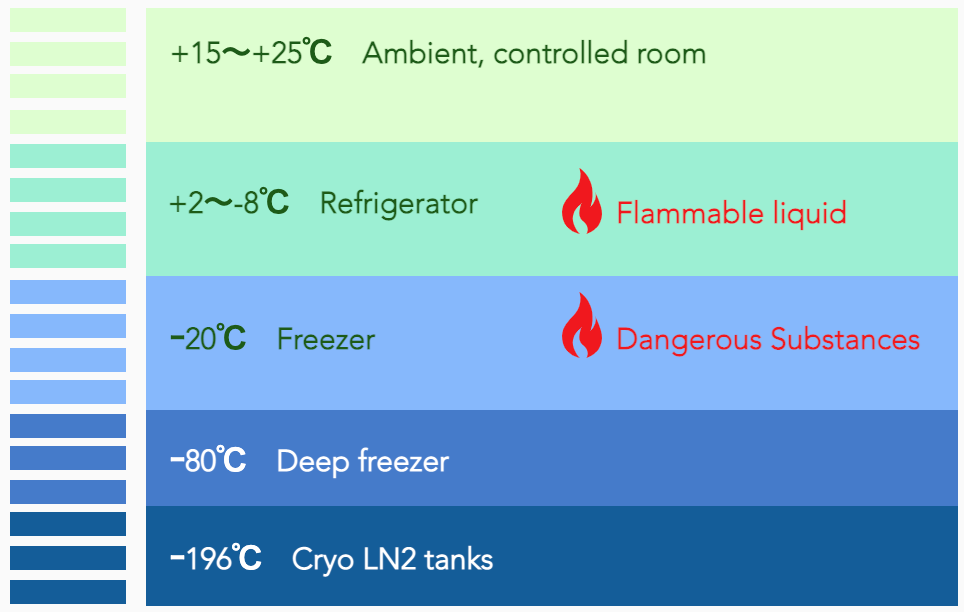

Pharmaceutical products

Reagents, whole blood
Plasma
Vaccine, Biological and cells
Regenerative medicine and cell
■Freezer chamber rental
Depending on the client requirement as well as frozen sample volume, GREEN8 provides individual freezer unit rental service. Clients can store frozen specimen by cryo-box unit, racks, or whole freezer unit as required.
■Temperature Measurement Report
Optional temperature monitoring reports can be generated during transportation from pickup site to storage facility, as well as temperature data logs during storage. Calibration of sensors and loggers are conducted.
Dangerous Goods storage and distribution
Dangerous Goods such as Japan Class IV explosive and flammable liquids can be stored in dedicated cold storage facilities at temperatures of around +5℃ or -20℃. GREEN8 emphasis on safety as well as compliance measures to provide proper handling of chemical and pharmaceutical products, according to Japan national and international standards.
Class 4 Dangerous Goods are:
Petroleum No. 1: Acetone, etc.
Petroleum No. 2: Chlorobenzene, etc.
Petroleum 3rd Class: Glycerin, etc.
Petroleum No.4 Bis Phthalate, etc.
Alcohols: Ethanol, etc.
※For handling other Japan DG substance categories,
please contact GREEN8 Japan for available service.
Cold storge temperature ranges
for Dangerous Goods:
+5 degree Celsius Chilled DG chamber
ー20 degree Celsius Frozen DG chamber

Hygiene and Sanitary Measures
■Strict hygiene measures
GREEN8 takes utmost care to implement GDP hygiene and sanitary measures. Our facility has measures to prevent insects and rodents’ entry and docking-shelter by installing sheet shutters, air curtains, and insect traps, as well as strict sanitary SOPs at the facility.
■Absolute Security
24 hours a day, 365 days a year security system, including a 24-hour CCTV surveillance system is implemented at GREEN8 facility in which access control to the warehouse is controlled through digital devices and equipped with alarm system. Our entrance security system keeps tracks of all the entries and exists of personnel, and prevents intruders or outsider’s access. Our strict security system prevents any leakage of internal information as well.
■Zoning management of biological and pharmaceutical material
GREEN8 facility implements zooning measures to handle pharmaceutical and biological substances. By separating the storage area into complete separate buildings, contamination and quality control is managed smoothly. Cell Culture Building is used for storage of medical and research specimens, whereas the Medical Supply Building is used for pharmaceutical raw materials and investigational drugs.


Disaster Prevention Measures
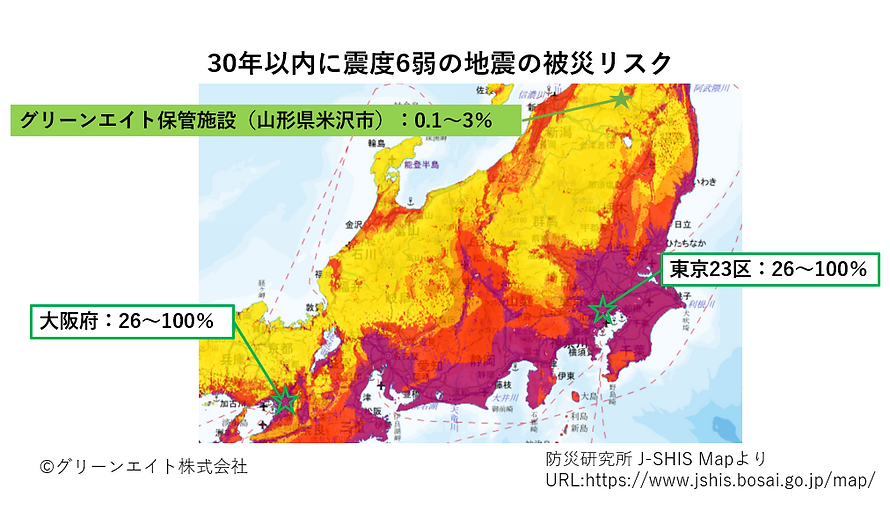
■Location with low risk of major earthquakes
GREEN8 Japan’s facility is located on land where the probability of an earthquake of intensity 6 or greater occurring in the last 30 years is between 0.1% to 3%. This risk analysis is extremely low compared to 26% to 100% risk of large-scale earthquake in Tokyo and Osaka metropolitans of Kanto and Kansai area. (Refer to Japan Seismic Hazard Information Station (J-SHIS) a public portal for seismic hazard information for Japan)。
■Stable Electricity Supply and Independent Emergency Generator
GREEN8 storage facility is supplied with electricity from two power grids, ensuring a stable power supply. In addition to this, the facility is equipped with a backup function using in-house power generation. In the event of a power failure, the cell culture and culture building can be supplied with electricity for 7 days, and the medical supply building for 3 days. We have constructed cold storage facility with complete backup systems for our customers' peace of mind in Japan.

Cargo acceptance details:
■Accepting bio-pharmaceutical materials such as :
Reproductive cells (frozen eggs, sperm, fertilized eggs, etc.) Biological specimens from human or animal origins Pathology specimens (cancer tumor specimens, etc.) Scientific specimens (genetically modified mouse cells, etc.) Chemicals substances (reagents, pharmaceutical raw materials, etc.) Dangerous materials (flammable substances, etc.) Pharmaceutical raw materials, intermediates, APIs
■Accepting storage conditions:
Cane
Vials (1ml or 2ml or other volume tubes)
Freeze box
Drum can
IBC container
Pallet
Corrugated cardboard
■Flexible storage period
From 1 month to long term
warehousing
We can provide a storage bio-pharmaceutical material flexibility from single 1 cane volume to pallets and IBC container volumes. Contact GREEN8 Japan team for costs and availability.
GREEN8 Japan Yonezawa Center
Yamagata, Japan
Click here for map
Total floor space: 405.80 m2
(Cell culture building: 220 m2,
Medical supply building: 250 m2)
Licenses and Permits:
Access:
1 km from Yonezawa Chuo IC of Tohoku Chuo Expressway
(about 2 min drive from nearest highway exit)
Close to National Route 13 (about 1 min.)
About 70 km from Yamagata Airport (about 70 minutes)
1.3 km from JR Yonezawa Station (about 3 minutes)
GREEN8 Japan Yonezawa Center is less than 4 hours one-way from the Tokyo metropolitan area. We contribute to safe and secure bio-pharmaceutical logistics service and to reduce your cost in Japan.

